
Small conformational changes have been seen, for example, upon formation of several different receptor/ligand complexes.ĭetermination of Protein Secondary Structure by Circular Dichroism: If there are any conformational changes, this will result in a spectrum which will differ from the sum of the individual components. Studying the conformational stability of a protein under stress - thermal stability, pH stability, and stability to denaturants - and how this stability is altered by buffer composition or addition of stabilizers and excipientsĬD is excellent for finding solvent conditions that increase the melting temperature and/or the reversibility of thermal unfolding, conditions which generally enhance shelf lifeĭetermining whether protein-protein or protein-ligand interactions alter the conformation of protein.
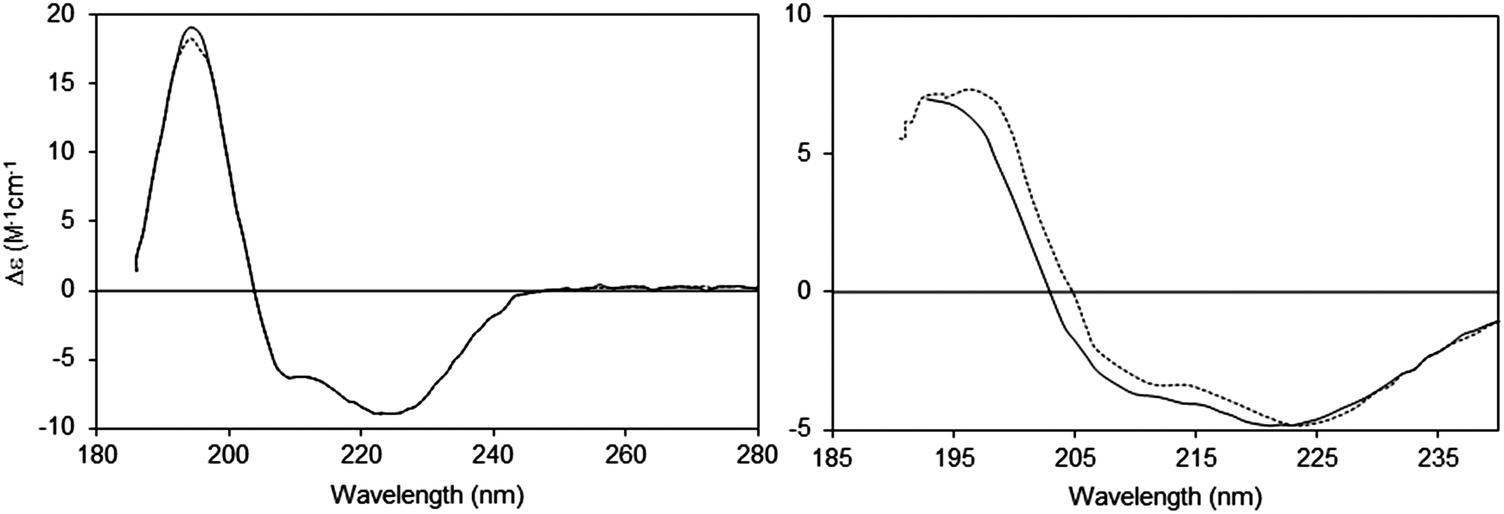
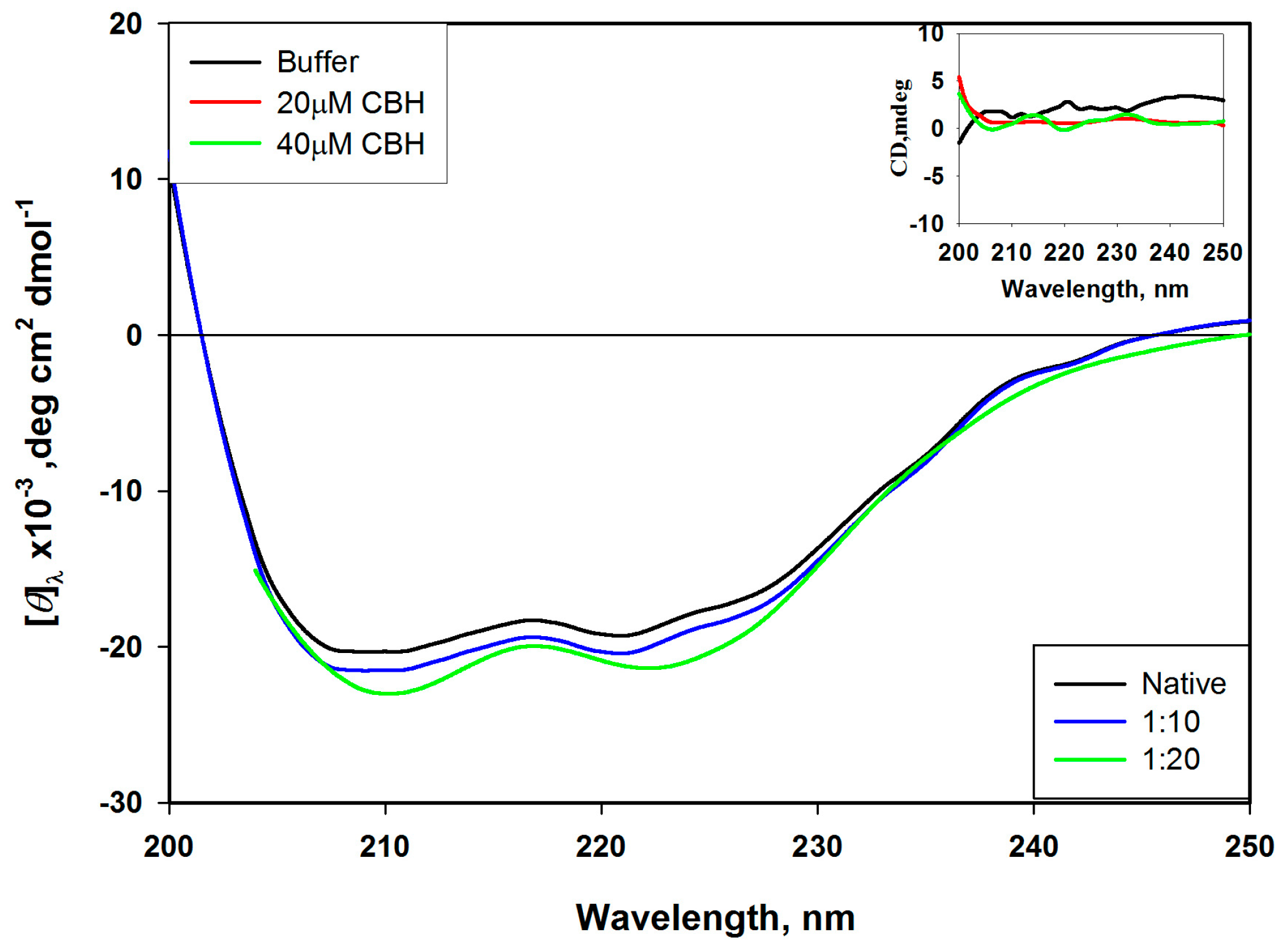
The absence of regular structure results in zero CD intensity, while an ordered structure results in a spectrum which can contain both positive and negative signals.Ĭircular dichroism spectroscopy is particularly good for:ĭetermining whether a protein is folded, and if so characterizing its secondary structure, tertiary structure, and the structural family to which it belongsĬomparing the structures of a protein obtained from different sources (e.g. species or expression systems) or comparing structures for different mutants of the same proteinĭemonstrating comparability of solution conformation and/or thermal stability after changes in manufacturing processes or formulation Circular dichroism (CD) spectroscopy measures differences in the absorption of left-handed polarized light versus right-handed polarized light which arise due to structural asymmetry.
